Posters
Posters
Smart Decision Making in Pharmaceutical Development Leveraging Automated Platforms
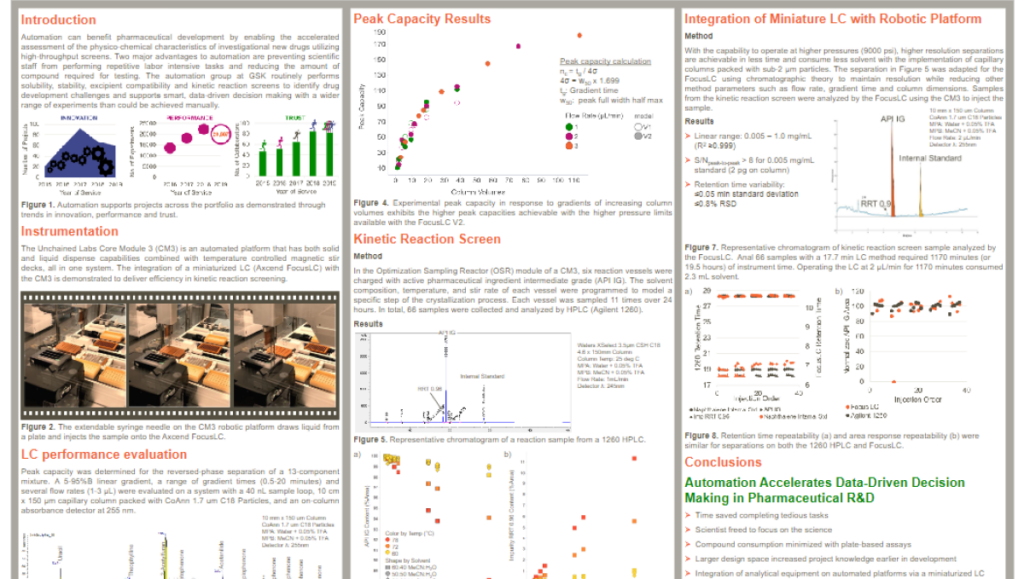
Automation can benefit pharmaceutical development by enabling the accelerated assessment of the physico-chemical characteristics of investigational new drugs utilizing high-throughput screens. Two major advantages to automation are preventing scientific staff from performing repetitive labor intensive tasks and reducing the amount of compound required for testing.
Targeted peptide quantification with small foot-print capillary LC-MS/MS
Quantification of low-abundance proteins remains a challenge for proteomics research. Standard LC-MS/MS often lacks the sensitivity to detect the crucial players in pathways, cellular processes, or biopharma manufacturing. Capillary LC-MS/MS emerges as a compelling solution, offering exceptional sensitivity and minimal sample volume requirements.
Interfacing Compact to Mass Spec
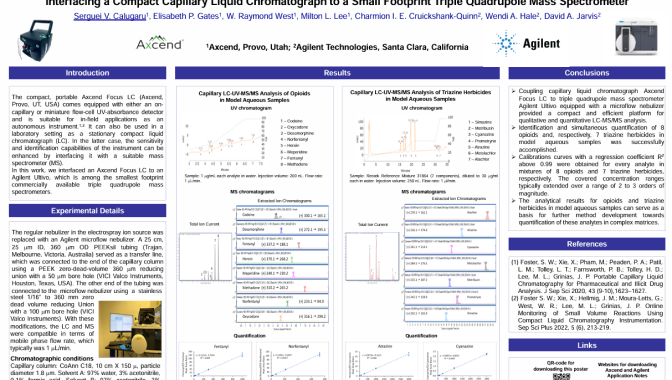
The compact, portable Axcend Focus LC (Axcend, Provo, UT, USA) comes equipped with either an on-capillary or miniature flow-cell UV-absorbance detector and is suitable for in-field applications as an autonomous instrument.1,2 It can also be used in a laboratory setting as a stationary compact liquid chromatograph (LC). In the latter case, the sensitivity and identification capabilities of the instrument can be enhanced by interfacing it with a suitable mass spectrometer (MS). In this work, we interfaced an Axcend Focus LC to an Agilent Ultivo, which is among the smallest footprint commercially available triple quadrupole mass spectrometers.
Have an application of interest?
Request App Note
Have an Application of interest that is not addressed in our Application Notes library? Please complete this form so we can learn more about your HPLC Application and investigate how we can work with you to get the results you need.
"*" indicates required fields